The larger the atom, the more electrons there are to move around, and the more volume they have to move around in. The size of the temporary fluctuating dipole you can get depends on the size of the atom. If the atoms were travelling slowly enough, they might stick together and start to form a liquid. The electrons in both atoms are, of course, still moving, and the nature of the dipoles will be constantly changing, but as long as the atoms were close together, they would change in synchronisation with each other. If another atom came close to this one, you would get a similar dipole induced in the second one, and they would attract each other. Where the electrons are densest there will be a slight negative charge, δ-, and the region around the nucleus a slight positive charge, δ+. 
That would give the atom an electrical dipole with the nucleus no longer at the centre of the electrons. Overall, the atom is electrically neutral.īut this is misleading, because it ignores the fact that the electrons are moving around within the atom, and at any one time they might be more unevenly arranged. You can picture one of their atoms as looking like this, with a positive nucleus at the centre of a number of negative electrons. That means that they go around as single atoms, not in pairs like oxygen and hydrogen, for example. Use the back button on your browser to come back here afterwards. Even if you have already read this, you need it fresh in your mind for the next bit. Important: Before you go on, it is essential that you read the page about intermolecular forces, particularly van der Waals dispersion forces. Nobody will expect you to remember these figures, but you might need to explain why they get bigger as the atoms get bigger. So all of these boil at temperatures well below room temperature. On this scale, a room temperature of 20☌ is 293 K. The temperatures quoted are all in Kelvin, the scale starting from absolute zero. Their boiling points increase with the size of the atom. 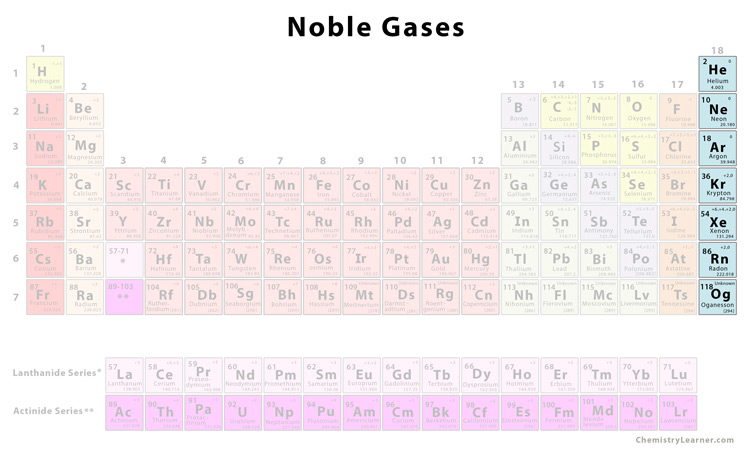
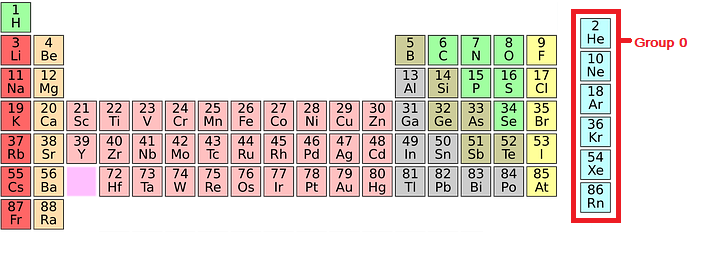
Obviously their name suggests that they are all gases. Radon is a radioactive gas produced from the radioactive decay of uranium and thorium. The modern name for Group 0 is Group 18, but I am using the older name for reasons I have already talked about on the page introducing the Periodic Table The main use of noble gases is to provide an inert atmosphere.įor example argon is used in lamps and light bulbs, as it will not react with the hot filament inside the bulb when it is hot.Īlso helium is used for filling balloons due to it’s low density compared to air.This page introduces the Noble Gases in Group 0 of the Periodic Table. The intermolecular forces become stronger so more energy is required to break these forces thus a higher boiling point as you go down the group. As you go down group 0, the atoms increase in size, as they have more electron shells. When atoms are boiled or melted, these intermolecular forces are broken. Between atoms of a particular element, there are weak attractive forces known as intermolecular forces. Explanation for change in boiling point.So helium has the lowest atomic mass and the lowest boiling point whereas radon has the highest atomic mass and the highest boiling point. As you go down group 0, the relative atomic mass increases and the boiling point of noble gases also increases. Chemistry Teacher Resource Edexcel | AQA | OCR.Biology Teacher Resource Edexcel | AQA | OCR | CIE.Booking A-Levels as a Private Candidate. 
Chemistry Flashcards Edexcel | AQA | OCR | CIE.Biology Flashcards Edexcel | AQA | OCR | CIE.Chemistry Textbook Edexcel | AQA | OCR | CIE.Biology Textbook Edexcel | AQA | OCR | CIE.Physics Weekly Classes Edexcel | AQA | OCR | CIE.Chemistry Weekly Classes Edexcel | AQA | OCR | CIE.Biology Weekly Classes Edexcel | AQA | OCR | CIE.GCSE Physics Flashcards Edexcel | AQA | OCR | CIE.Physics Online Course Edexcel | AQA | OCR | CIE.Chemistry Online Course Edexcel | AQA | OCR | CIE.Biology Online Course Edexcel | AQA | OCR | CIE.Physics Tutor Edexcel | AQA | OCR | CIE.Henrietta Barnett School Admissions Tutors.James Allen’s Girls’ School Admissions Tutors.King’s College Wimbledon School Admissions Tutors.Lycee Francais Charles de Gaulle School Admissions Tutors.Radley College School Admissions Tutors.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
